Isolating VEGF was merely one step. Next, Ferrara and his team wanted to see how important the factor was. To do this, the scientists knocked out the VEGF gene in mice. “It showed that even if you inactivated one of the two alleles, there is embryonic lethality,” said Ferrara. “This showed that this was a very important factor, not easily replaced.” Ferrara’s group, along with other scientists at the University of California at San Francisco, then isolated a receptor called FMS-like tyrosine kinase 1 that bound to VEGF with a very high affinity (9). As researchers continued to unravel VEGF biology, they isolated other members of the VEGF family, including VEGF-B, C, and D.
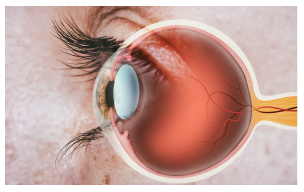
Meanwhile, scientists who studied the eye became very interested in how VEGF might fit into the progression of ocular diseases, some of which are caused by markedly increased blood vessel growth in the eyeball. One of them was Joan Miller, an ophthalmologist at Harvard Medical School who had just joined Folkman’s lab during her retina fellowship.
During her training, Miller developed nonhuman primate models of eye diseases. By using these models and studying retinal cells in a dish, Miller and the team found that increased levels of VEGF correlated with increased vascularization in the eye — and that, specifically, it was the retina that produced VEGF (10,11). Finally, the scientists showed that “you could take VEGF protein and inject it into the vitreous of normal animal eyes and develop new blood vessels,” Miller said. “So it was sufficient in and of itself to produce new blood vessels” (12).
Miller’s findings were recapitulated by a larger study showing that in human patients, VEGF was indeed associated with the growth of new blood vessels (13). With these results in hand, scientists began to test Folkman’s original 1971 theory — if blocking VEGF would in fact reduce tumor growth or, with the new knowledge that the molecule was heavily implicated in the eye and ocular diseases.